When DeepMind's AlphaFold transformed protein structure prediction in 2020, it was rightly treated as a major scientific breakthrough. Researchers could suddenly predict the three-dimensional structure of many proteins from sequence with useful accuracy, and large public structure resources expanded dramatically.
But predicting a protein's structure is only the beginning. The drug discovery question is more practical: how do you use that structure to design a medicine that works in living biology?
From Structure to Drug Design
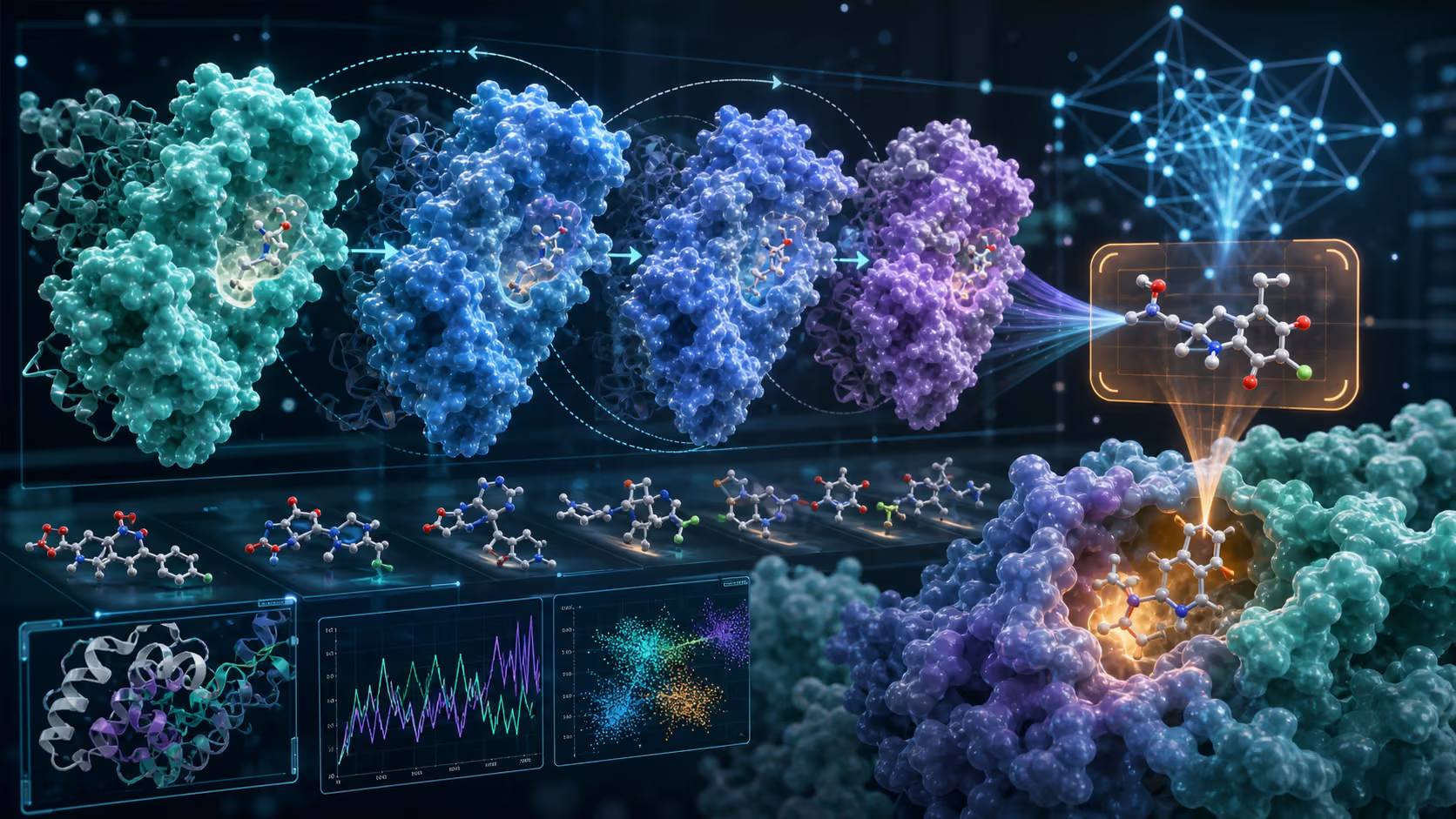
Knowing a protein's shape tells you where a drug molecule might bind. These binding sites—pockets on the protein surface—are where small molecules can attach and modulate the protein's function. Identifying and characterizing these pockets is the critical first step in structure-based drug design.
Traditional approaches relied on X-ray crystallography and cryo-EM to determine binding sites experimentally—a process that could take months or years. AI-predicted structures accelerate early reasoning, but they also introduce a new responsibility: knowing when a predicted model is good enough for design and when experimental structure is still needed. Proteins in the body are dynamic, constantly shifting between conformations.
Dynamic Structural Intelligence
The next frontier in AI-driven drug design goes beyond static structure prediction. It involves understanding the dynamic behavior of proteins:
- Conformational ensembles: Proteins sample multiple shapes. A drug that binds to a rare conformation might be more selective and effective than one targeting the most common shape.
- Allosteric sites: Some of the most promising drug targets are sites distant from the active site that modulate protein function through conformational change.
- Protein-protein interactions: Many diseases involve aberrant protein partnerships. Disrupting these interactions requires understanding the dynamic interface between two proteins.
AgentCures incorporates these dynamics into its molecular design process. Our AI agent doesn't just dock molecules into a static pocket—it evaluates how molecular candidates interact with the full conformational landscape of the target protein.
Structures Are Starting Points
A structure can reveal a pocket, suggest a scaffold, or explain why one molecule is selective and another is not. It cannot, by itself, prove that a drug will be safe, bioavailable, or clinically useful. The most valuable systems combine structure with ADMET prediction, disease biology, assay data, and patient context.
Integrating Structure Into the Pipeline
What makes AgentCures' approach distinctive is that structural intelligence is not a separate step—it is woven into every stage of the drug discovery pipeline:
- Target identification: AI analyzes protein structures to identify druggable targets that others might overlook.
- Hit generation: Generative models design molecules shaped to fit specific binding pockets.
- Lead optimization: Structure-activity relationships are continuously updated as the AI agent learns from each round of virtual screening.
- Selectivity engineering: Structural comparisons across protein families help the agent design molecules that hit the intended target without affecting related proteins.
The Structural Data Advantage
The explosion of structural data—from both experimental and AI-predicted sources—has created an unprecedented opportunity. Companies that can effectively mine this data will identify drug targets and design molecules that would be invisible to traditional approaches.
AgentCures processes structural data at scale, integrating it with genomic, transcriptomic, and clinical data to build a comprehensive picture of each disease target. This multi-modal approach is what enables our AI agent to make design decisions that account for biological complexity rather than treating drug design as a simple lock-and-key problem.
What This Means for Patients
Ultimately, better structural understanding translates to better drugs. Molecules designed with structural intelligence are more likely to be selective (reducing side effects), potent (requiring lower doses), and effective (reaching the right target in the right conformation). For patients, this means safer, more effective treatments arriving faster than ever before.
The protein structure revolution did not end with AlphaFold. It began there, and the next phase is turning structural insight into better medicines.